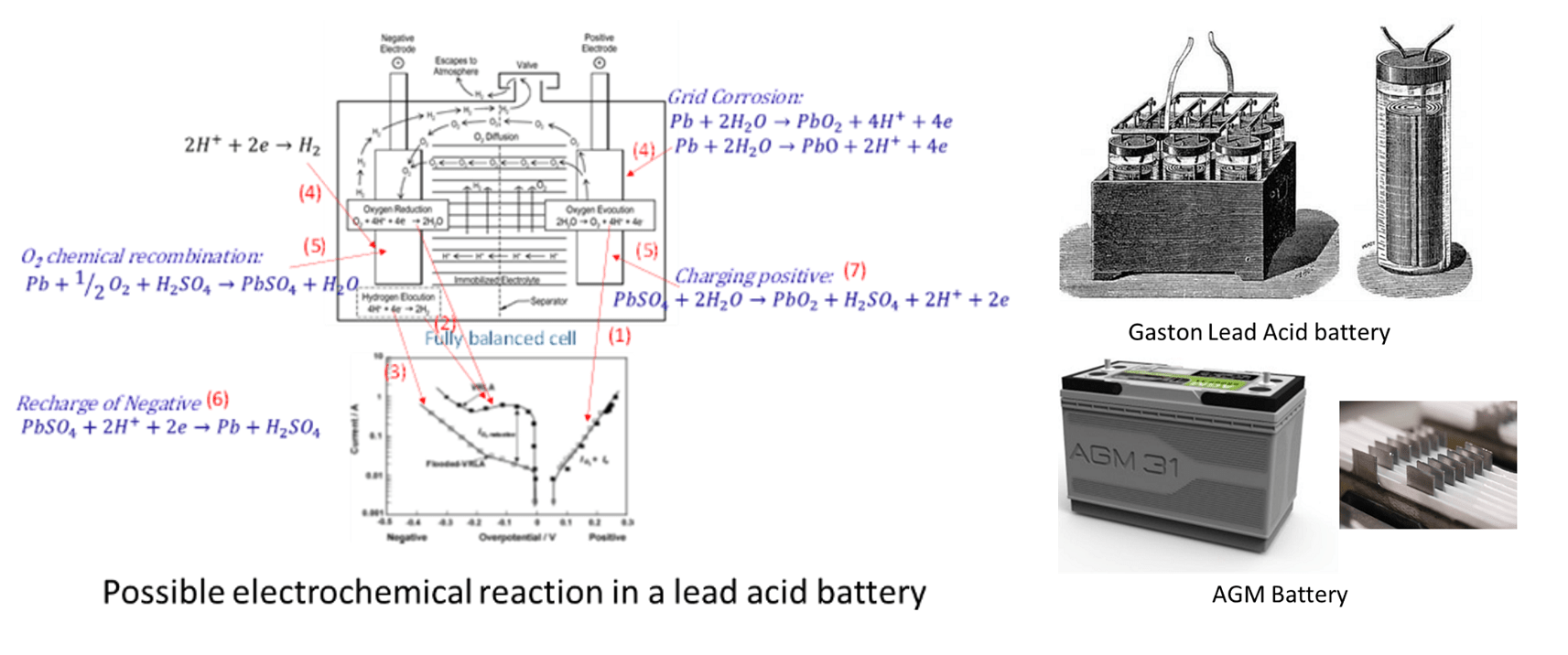
Since Gaston Plante, for the first time demonstrated a rechargeable Lead-acid battery in front of French Academy of Science on March 26th, 1860, Lead-acid batteries have been a work horse to provide portable energy in almost every areas in our life. Only until in the past a few decades, rechargeable Ni-Cd, Metal –hydride and now the state-of-art Li-ion batteries are gradually replacing lead acid in many applications, in which high energy density and high portability are essential e.g. cell phone, lab top computer, hybrid and plug-in electric vehicles.
The largest markets for lead acid batteries are SLI battery (starting, lighting and ignition) for the vehicle engines and storage battery e.g. for UPS (uninterruptible power system). The technology advancement had been stagnant till the quick proliferation of Start n’ Stop application in the internal combustion vehicle. The advanced AGM (Absorbent Glass Mat) and EFB (Extended Floored Battery) started to surface especially in past a few years. The AGM design aimed to mitigate the following three shortcomings for a lead acid battery.
- Stratification: When a flooded lead acid battery is discharged, sulfuric acid is consumed and water is produced in the positive plates. The less density water forms at upper portion of electrolyte reservoir. During charge, however, pure sulfuric acid is produced and sinks to the bottom portion of the electrolyte reservoir. The development of a vertical concentration gradient of sulfuric acid was called “electrolyte stratification”.
- Sulfation: During the discharge, PbSO4 start to form on the porous electrodees of membrane sizes between them. PbSO4 crystal becomes larger and the surface areas of electrode become smaller, while tet-PBO starts to form. The process is sulfation. It occurs on the negative plate.
- Dynamic Charge Acceptance: